Salivary Cortisol and the Laboratory Evaluation of Cushing’s Syndrome
Normally, the control of cortisol production involves the interaction of the hypothalamus, anterior pituitary gland and the adrenal cortex.
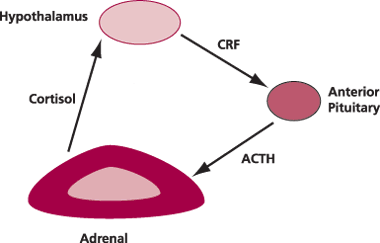 |
| Feedback mechanism for cortisol regulation. The hypothalamus secretes corticotropin releasing factor (CRF) which stimulates the production of adrenocorticotropin (ACTH) by the anterior pituitary gland. ACTH stimulates production of cortisol by the adrenal cortex. |
Cushing’s syndrome results from overproduction of cortisol by the adrenal cortex. It is a complex disease because it may be caused by 4 different problems.
| 1. | Iatrogenic—administration of steroids for immunosuppression in the treatment of autoimmune diseases, other inflammatory conditions such as Crohn’s disease, and as part of immunosuppression for transplantation. |
| 2. | Pituitary hypersecretion of adrenocorticotropin (ACTH). This accounts for 75-80% of cases of non-iatrogenic (endogenous) Cushing’s syndrome. The usual cause is an adenoma of the basophilic cells in the anterior pituitary gland, although chromophobe adenomas have also been shown to secrete ACTH. In this case, the adrenal glands are bilaterally hyperplastic, ACTH levels are usually elevated in the serum along with levels of cortisol. About 80% of these lesions are most frequently found in young, adult females. |
| 3. | Rare Ectopic ACTH production (or production of corticotropin releasing Factor—CRF) by nonpituitary tumors. These have been most frequently associated with small cell carcinoma of the lung, carcinoids and other small cell neoplasms. These lesions are most often detected in late middle-aged men. |
| 4. | Primary adrenal hyperproduction accounts for the remaining 15-20% of cases. These are due to either unilateral adrenocortical tumors (benign or malignant) or bilateral adrenal hyperplasia. In adults, the occurrence of adenomas and carcinomas is about equal, whereas in children, adrenal carcinomas are more frequent. Bilateral autonomous hyperplasia is uncommon. Carcinomas tend to produce higher levels of serum cortisol than do adenomas or bilateral autonomous hyperplasia. Because the anterior pituitary is under feedback control of the level of cortisol, the normal production of ACTH by the anterior pituitary is suppressed. As a result, the “non-hyperplastic” parts of the adrenal cortex will be suppressed. From the biochemical diagnostic perspective, however, all of these cases present with elevated levels of cortisol and low levels of ACTH. |
Pathophysiology
Normally, the ACTH stimulation of cortisol occurs in a circadian fashion such that cortisol has much higher levels early in the morning, while late in the evening the levels are low. The salivary cortisol assay takes advantage of the normally low levels of cortisol in the evening. However, it may be difficult to distinguish between early Cushing’s syndrome and some conditions of stress because the latter result in an increased production of cortisol.
Clinically, the excess production of cortisol results in central (often referred to as “truncal”) obesity, excess fat in the face and supraclavicular fat pads in the upper back (forming a “buffalo hump”). Other major effects from excess cortisol include: thinning of the skin, easy bruising, characteristic purple abdominal striae, atrophy of proximal muscles, gonadal dysfunction, psychological problems, hypertension, atherosclerosis, congestive heart failure, osteoporosis, predisposition to infections and poor wound healing.
Biochemically, in addition to the abnormal ACTH and cortisol values, there is an abnormal glucose tolerance test in about 90% of patients with Cushing’s syndrome. Hypokalemia, though only found in about a quarter of these patients, has 95% specificity for Cushing’s syndrome.
Laboratory Testing for Cushing’s Syndrome
The best currently available screening test for Cushing’s syndrome is the Salivary Cortisol test (discussed below).
Formerly, the best screening test for excess production of cortisol was the 24-hour urine for urinary free cortisol and creatinine. Another test, still commonly used today, is the Dexamethasone suppression test. We no longer recommend using either of these as the first line screening test. The salivary cortisol test has a higher sensitivity and specificity. However, because these other tests are still commonly used they will be discussed briefly.
Twenty-four hour urinary free cortisol (UFC)
Principle — UFC provides a measure of free (unbound) cortisol that had circulated in the blood for the 24-hour period. The advantage of this test is that it is not affected by factors that influence corticosteroid-binding globulin levels. Further, because of the circadian changes, the 24-hour test gives a better overview of the total amount of cortisol secreted than does a single plasma cortisol.
Unfortunately, there are some problems that occurr with this assay.
| 1. | Any 24-hour collection of urine is a clumsy specimen because if the patient misses some collections, the results will be inaccurate. |
| 2. | It could be falsely negative if the production of cortisol is intermittent. Therefore, when the UFC test was negative, we recommended that it should be repeated up to three times. If all three are normal, the diagnosis of Cushing’s syndrome is considered “highly unlikely.” |
| 3. | In order for the UFC test to be valid, renal function must be normal. This is why we perform a urinary creatinine level to document both the adequacy of the collection and the basic level of renal function. |
| 4. | Another problem with the assay is the interference by prednisone. |
Interpretation — UFC values that were greater than four times the upper limit of normal are rare except in Cushing’s syndrome and are considered diagnostic. Unfortunately, milder elevations are not uncommon, being found in alcoholism, depression, chronic anxiety, and normal pregnancy.
Specimen Required — The submitting laboratory should provide a 20 ml aliquot of the 24-hour urine that has been collected in the presence of a preservative (usually boric acid—10 gm/L) or by keeping the urine at 4ºC. In order for us to properly calculate the total cortisol, we need to be provided with the total volume of the sample. Because of the circadian rhythm of normal cortisol levels and responses to stress, we do not perform the UFC test on random urine samples. A refrigerated sample could be processed up to 7 days. If the sample needs to be stored longer, it should be frozen (which allows processing up to one month).
| The Reference Range for the UFC Test | |
| Urine Free Cortisol: | 8-77 mcg/24 hours |
| Urine Creatinine: | Male: 1.0-2.0 g/24 hours Female: 0.8-1.8 g/24 hours |
Low-dose dexamethasone suppression test (DST)
Principle — Dexamethasone is a potent suppressor of ACTH, yet it does not cross-react with the assay for cortisol. In patients with a normal hypothalamic-pituitary-adrenal feedback, it will suppress production of CRF, ACTH and cortisol. However, patients with any of the 3 forms of endogenous Cushing’s syndrome (above) will not undergo suppression because of the autonomous function of the affected organ.
To perform the test, 1mg of dexamethasone is given orally between 11 p.m. and midnight. The fasting serum cortisol level is measured the next morning between 8 a.m. and 9 a.m. A major advantage is the ease with which it can be performed in the outpatient setting.
Specificity of the test is limited due the complexity of serum (or plasma) studies that include not only free but cortisol bound to cortisol binding globulin. In addition, failure to suppress can occur in apparently healthy individuals who do not reach the cut off of 5 mcg/dL. This is most frequently found in patients who do not have Cushing’s syndrome, but who suffer from other illnesses associated with stress (acute and chronic infections), patients with alcohol abuse and those with depression. The DST has a false positive rate of about 30% in these individuals. Since those conditions are much more common than Cushing’s syndrome, a positive DST is usually a false positive test.
Furthermore, some patients with Cushing’s syndrome do have the serum cortisol levels falling below 5 mcg/dL, limiting the sensitivity of the findings.
Interpretation — The original criterion for normal level of suppression is a cortisol level below 5 mcg/dL (138 nmol/L). Some recent studies suggest that by lowering this to 1.8 mcg/dL (50 nmol/L) it improves the sensitivity of detecting mild hypercortisolism. If the serum cortisol is suppressed below 1.8 mcg/dL, it excludes active Cushing’s Syndrome at that time.
Specimen Required — We require 1 serum separator tube, preferably with 0.5 mL of serum (0.3 mL minimum). The specimen should be drawn between 7 a.m. and 9 a.m. the morning after the administration of dexamethasone. Serum should be separated from the clot within one hour of obtaining the sample and it should be and refrigerated. Refrigerate 72 hours or freeze.
Late-night salivary cortisol
The most sensitive and best screening test for Cushing’s syndrome is the late-night salivary cortisol test.
Principle — Cortisol in saliva correlates well with serum (plasma) cortisol, independent of salivary flow rates. Free cortisol diffuses through acinar cells of the salivary glands into the mucosal surface of the oral cavity and only a small fraction (about 15%) is bound to cortisol binding globulin. Salivary cortisol is in equilibrium with the free cortisol of the serum. Because it reflects the unbound, biologically active form of serum cortisol, it does not have the problems with variation in level of cortisol binding globulin that serum tests suffer from. Another advantage of this test is that cortisol in saliva is stable at room temperature for up to a week. A salivette device is provided by the laboratory that allows the patient to conveniently take their own sample at home at 11 p.m. This test has been found to work well with adults, children and in patients with cyclical Cushing’s syndrome.
This test has been reported to provide as much as 100% specificity with 91-93% sensitivity for Cushing’s Syndrome. The 7-9% of cases that were missed in one study gave results in the “pseudo-Cushing’s Syndrome” range. Papanicolaou et al. reported the diagnostic value of nighttime salivary cortisol measurements was similar to that of serum cortisol measurements taken at the same time.
| Reference Ranges (established by the reference laboratory) | |
| Prepubertal Children: | Midnight values <0.05-0.19 mcg/dL |
| Adults: | 11:00 PM values 0.05-0.17 mcg/dL |
References
- Arnaldi, G. et al., Diagnosis and complications of Cushing's Syndrome: a consensus statement. J Clin Endocrinol & Metabol 88;593,2003.
- Findling, JW et al., The low-dose dexamethasone suppression test: a reevaluation in patients with Cushing's Syndrome. J Clin Endocrinol & Metabol 89;1222,2004.
- Isidori AM. Et al., Discriminatory value of the low-dose dexamethasone suppression test in establishing the diagnosis and differential diagnosis of Cushing's Syndrome. J Clin Endocrinol & Metabol 88;5299,2003.
- Papanicolaou, DA et al., Nighttime salivary cortisol: a useful test for the diagnosis of Cushing's Syndrome. J Clin Endocrinol & Metabol 87;4515,2002.
- Van Aken, MO et al., Automated measurement of salivary cortisol. Clin Chem 49;1408,2003.

