Vitamin B1 Thiamine

Introduction
Vitamin B1 (Thiamine) is a water-soluble vitamin of the B group. In animal tissues, it is present mainly in phosphorylated form, with thiamine pyrophosphate being the most abundant. The pyrophosphate form is a coenzyme of five different enzymes involved in carbohydrate and lipid metabolism. Vitamin B1 is absorbed by the small intestine by a specific, easily saturable mechanism, which can be impaired by alcohol, and by synthetic or natural thiamine antagonists present in food.
Thiamine deficiency can develop after only one month of a thiamine free diet. Mild thiamine deficiency occurs in pregnant women (increased requirement), in alcoholics, the elderly, persons who have persistent vomiting or gastric suction, those with chronic illnesses such as cancer, patients taking diuretics, and those who go on a long fast. Severe thiamine deficiency is called beriberi and is characterized by peripheral neuritis or cardiac failure. In undeveloped countries, the main cause of severe thiamine deficiency is inadequate intake due to a diet consisting primarily of polished rice. In industrialized countries, however, the most common cause of severe thiamine deficiency is alcoholism.
Chemistry
The structure of thiamine (vitamin B1) (3-[4-amino-2- methyl-pyrimidyl-5-methyl]-4-methyl-5-[β-hydroxy-ethyl] thiazole) is that of a pyrimidine ring, bearing an amino group, linked by a methylene bridge to a thiazole ring. The thiazole ring has a primary alcohol side chain, which can be phosphorylated in vivo to produce thiamine phosphate esters, the most common of which is thiamine pyrophosphate (Figure 1). Monophosphate and triphosphate esters also occur.
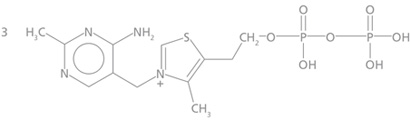
Figure 1. Thiamine pyrophosphate
Dietary Sources
All plant and animal tissues contain vitamin B1, and so the vitamin is present in all natural unprocessed foods. Rich sources of vitamin B1 include yeast, pork, legumes, beef, whole grains, oatmeal, and nuts. Milled cereals and milled and polished rice contain little thiamine, if any (1). Thiamine deficiency is therefore more common in cultures that rely heavily on a rice-based diet.
Certain foods such as tea, coffee, raw fish, and shellfish contain thiamineases, which can destroy the vitamin. Thus, drinking large amounts of tea or coffee can lower thiamine body stores (2).

Biological Functions
The biologically active form of thiamine is thiamine pyrophosphate (Vitamin B1, thiamine diphosphate). It functions as a co-enzyme (a co-carboxylase) in the oxidative decarboxylation of pyruvate to form acetate and acetyl co-enzyme A, and in the oxidative decarboxylation of alpha-keroglutarate to succinyl Co A. Blocking the first metabolic reaction results in impaired nerve transmission. Blocking the second reaction results in anaerobic metabolism with subsequent increases in blood pyruvate and lactate.
Thiamine pyrophosphate also functions as the coenzyme for the enzyme transketolase, an enzyme involved in the pentose monophosphate pathway. This metabolic pathway supplies pentose phosphate for nucleotide synthesis, as well as reduced nicotinamide adenine dinucleotide phosphate (NADPH), an important electron donor in reductive biosynthesis.
Thiamine also has a non-coenzymatic role in nerve function as is evidenced by signs of thiamine deprivation which are mainly neurological. The biochemical nature of this role is still unclear.
Mild Deficiency
Although severe thiamine deficiency is rare today, large segments of the world’s population continue to subsist on marginal or sub-marginal intake of thiamine. People exposed to subclinical thiamine deficiency are predisposed to manifest severe deficiency under appropriate circumstances.
Mild thiamine deficiency can be seen in people who have high carbohydrate intakes and low thiamine intakes e.g. in people whose staple food is polished rice, especially if their diet contains anti-thiamine factors (tea, coffee, raw fish) and in population groups who consume large quantities of sweetened carbonated drinks and candies (3).
Groups whose minimum thiamine needs are markedly increased because of increased metabolic demand are also at risk and include pregnancy and lactation, intercurrent illness (cancer, liver disease, infections, hyperthyroidism), surgery, and whenever thiamine absorption is reduced by regular high alcohol intake, gastrointestinal disease, dysentery, diarrhea, or nausea/vomiting (4).
Mild deficiency may also be seen in patients with congestive heart failure treated with diuretics (5, 6, 7). Recently it has been suggested that patients with Alzheimer’s disease may be thiamine deficient (8, 9, 10), but these studies are weak and contradictory. Whether thiamine supplementation is of benefit in Alzheimer’s Disease remains controversial.
Characteristic early symptoms of thiamine deficiency appear after 2 to 3 weeks of a deficient diet and include anorexia, irritability, fatigue, aching, burning sensation in the hands and feet, indigestion, sleep disturbances, and depression. After 6 to 8 weeks the only objective signs at rest may be a slight fall in blood pressure and moderate weight loss. After 2 to 3 months apathy and weakness become extreme, calf muscle tenderness develops with loss of recent memory, confusion, ataxia, and sometimes persistent vomiting (3).

Severe Deficiency
Severe thiamine deficiency is called beriberi. Three main types are recognized: “dry” or neuritic, “wet” or edematous, and “infantile” or acute.
Dry Beriberi
Dry beriberi is a disease of the peripheral nervous system involving bilateral impairment of sensory, motor, and reflex functions. The neuropathy begins in the feet and legs and then extends up the body. Early signs of dry beriberi often include sensations of pins and needles and numbness in the feet. The legs, especially the calves, feel heavy and weak so that walking becomes uncomfortable. As the disease progresses, there is a marked wasting of the leg muscles and even slight pressure applied to the calves elicits severe pain. The characteristic foot and wrist drop develop and there may be complete flaccid paralysis of the lower, and occasionally upper, extremities (1).
Wet Beriberi
In wet beriberi, thiamine deficiency affects the cardiovascular system by causing arteriolar dilation throughout the circulatory system and by weakening the heart muscle. Physical signs of wet beriberi are indicative of high output cardiac failure; they include tachycardia, rapid circulation time, elevated peripheral venous pressure, and widespread edema.
Infantile Beriberi
Infantile beriberi is rarely seen today but can occur in breast-fed babies between the second and sixth month of life. Vomiting is one of the most important early signs of infantile beriberi. Other symptoms include constipation, agitation, difficulty in breathing, and generalized edema (1). In severe cases the child appears to be crying but no sound is heard or only a thin whine. This characteristic feature, aphonia, is due either to paralysis of the laryngeal nerve or to edema of the vocal cords. A chronic, pseudomenningial form of beriberi which affects the central nervous system and may cause convulsions occurs in older infants aged 7 to 9 months.

Wernicke-Korsakoff Syndrome
The Wernicke-Korsakoff syndrome is a thiamine deficiency disease associated with chronic alcoholics who obtain more than half their daily calories from ethanol. Binge drinkers who eat normally between bouts are not prone to the disease. The Wernicke-Korsakoff syndrome has two components, Wernickes’ disease and Korsakoffs’ psychosis. Wernickes’ disease is characterized by a triad of clinical signs – eye abnormalities, incoordination, and altered state of consciousness. Korsakoffs’ psychosis is characterized by a form of amnesia in which events of ordinary life are forgotten as quickly as they occur but the events in the distant past are well remembered. Another symptom, confabulation (story-telling) is an attempt by the patient to hide the amnesia.

The Determination of Thiamine Status
Thiamine status can be assessed by three types of tests: determination of erythrocyte transketolase activity, the urinary excretion of thiamine before and after thiamine administration, and serum, erythrocyte, or whole blood thiamine levels.
Erthyrocyte transketolase activity is usually measured by measuring the rate of disappearance of D-ribose-5-phosphate, in the presence of orcinol reagent, before and after thiamine pyrophosphate (Vitamin B1) supplementation. The percent increase in activity is defined as the “TPP effect.” The transketolase determination was onceconsidered the most reliable means of assessing thiamine status, but is now considered inadequate due to non-specificity, poor sensitivity, poor precision, and lack of specimen stability.
The determination of the urinary excretion of thiamine in a 4-hour specimen, before and after a 5 mg dose of thiamine, has been used as a measure of thiamine status. However, excretion of the water soluble vitamin can be influenced by dietary intake, absorption, and other factors. Because of these factors, and the inconvenience of sample collection, this test is now considered obsolete.
Thiamine levels can be determined in serum or plasma, but this method suffers from poor sensitivity and specificity and serum thiamine levels reflect recent intake rather than an assessment of total body stores of thiamine.
Since erythrocyte thiamine stores deplete at a similar rate to other major organs (11) and since the erythrocyte contains approximately 80% of the total thiamine content of whole blood (12), mainly as the pyrophosphate, the analysis of thiamine pyrophosphate in whole blood or erythrocytes is the most appropriate measurement to assess thiamine status.
Whole blood thiamine levels are determined, at Warde Medical Laboratory, by oxidizing thiamine and its phosphate esters to their corresponding thiochrome derivatives. Thiochrome diphosphate (Figure 2) is selectively measured by separation on an HPLC column followed by fluorometric detection. This method is sensitive and specific and it measures only the active form of thiamine: thiamine diphosphate (Vitamin B1).
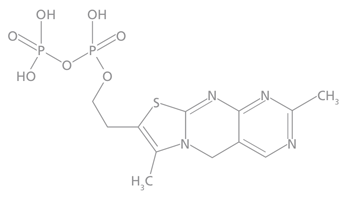
Figure 2. Thiochromediphosphate
Summary
Thiamine (Vitamin B1) is an essential vitamin required for carbohydrate and lipid metabolism. Thiamine is found in many foods so deficiency develops primarily due to poor nutrition. The symptoms of mild deficiency are vague but include loss of appetite, irritability, fatigue, and sleep disturbances. Severe deficiency occurs in undeveloped countries due to diets consisting primarily of processed rice, and in developed countries as a result of alcoholism. The most reliable laboratory test for thiamine deficiency is the measurement of thiamine pyrophosphate in whole blood.
References
-
- G.F.M. Ball, Vitamins: Their Role in the Human Body. p274, Blackwell Publishing, Malden, MA (2004).
-
- Russell RM, Vitamin and Trace Mineral Deficiency and Excess, in Harrison’s Principles of Internal Medicine, 16th edition, McGraw-Hill, New York (2005).
-
- Anderson SH, Charles TJ, Nicol AD. Thiamine Deficiency at a District General Hospital: Report of Five Cases. Quarterly Journal of Medicine, 1985, 55: 15-32.
-
- Thiamine Deficiency and Its Prevention and Control in Major Emergencies, World Health Organization, 1999.
-
- Brady JA, Rork CL, Horneffer MR, J. Am. Diet. Assoc. 95 (1995) 541.
-
- Shimon I, et. al., Am. J. Med 98 (1995) 485.
-
- Seligmann H, et. al., Am. J. Med 91 (1991) 151.
-
- Gold M, Chen MF, Johnson K, Arch. Neurol. 52 (1995) 1081.
-
- Meador K, et. al., J. Geriatr. Psychiatry Neurol. 6 (1993) 222.
-
- Mastrogiacoma F, et. al. Neurol. 39 (1996) 585.
-
- Brin M, Erythrocyte as a Biopsy Tissue for Functional Evaluation of Thiamine Adequacy. JAMA 1964; 187: 762-6.
- Schrijver J, et. al., A Reliable Semiautomated Method for the Determination of Total Thiamine in Whole Blood by the Thiochrome Method with High-Performance Liquid Chromatography. Ann Clin Biochem 1982; 19: 52-6.

